Visualize trends, 3D orbitals, isotopes, and mix compounds. You will see in the next chapters that the chemical properties of elements are determined by the number of valence electrons. Interactive periodic table showing names, electrons, and oxidation states. This arrangement is emphasized in Figure 6.29, which shows in periodic-table form the electron configuration of the last subshell to be filled by the Aufbau principle.

It is in period 4 and family 15 (5A or the Nitrogen family)., Two representative elements are in the same period of the periodic table. We can see from the electron configuration of a carbon atom-1 s 22 s 22 p 2-that it has 4 valence electrons (2 s 22 p 2) and 2 core electrons (1 s 2). Now we can understand why the periodic table has the arrangement it hasthe arrangement puts elements whose atoms have the same number of valence electrons in the same group. Study with Quizlet and memorize flashcards containing terms like How many valence electrons does each atom of arsenic (As) have Arsenic is element 33. Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. Thus, it is convenient to separate electrons into two groups. The periodic table shows the number of electrons in the valence shell of a chemical element that are available in the formation of a chemical bond. Fluorine is one of the lightest members of the halogen elements. With this printable periodic table with valence electrons you can check how many electrons a chemical element has in it’s outermost electron shell. This reacts with other elements but not with argon, neon, and helium. As compared to the other electronegative element, it is totally reactive in nature.
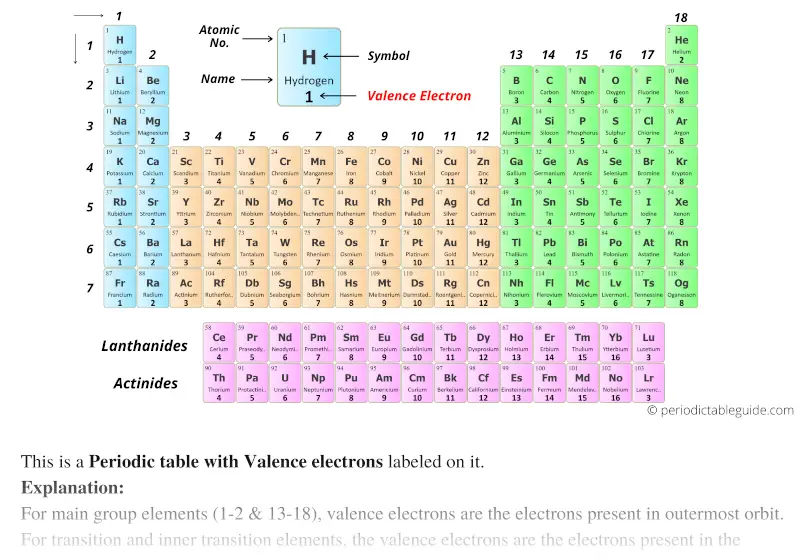
The path a specific element will take depends on where the electrons are in the atom and how many there are. Fluorine Valence Electrons: Fluorine is a chemical element that has a symbol F. For main group elements, the number of valence electrons usually ranges between 1 and 8 because eight electrons forms a complete octet. \,3s^1 \nonumber \]Ī chemical reaction results from electron removal, electron addition, or electron sharing of the valence electrons of the different atoms.
